If you were to walk into a hospital in the United States and ask for the Substance Abuse Clinic at the front desk, you will probably get a funny look.
There are areas of medical practice for kidney disorders, heart disorders, gastro-intestinal disorders, and every other organ system in the body. There are even plenty of medical fields dealing with the brain: neurology, neuropathology, neurosurgery, psychiatry, etc. Indeed, medical training specifically for treatment of substance abuse disorders is reserved largely for the field of psychiatry.
This seems odd since substance abuse is one of the most pervasive public health problems in the country[a] and in fact, the Centers for Disease Control and Prevention has recently declared the prescription opioid painkiller and heroin problem a national epidemic[b].
So, what gives? Here, we’ll explore the brain and the neuroscience of addiction…from the perspective of a neuroscientist! Then, we invite your feedback and comments at the end.
Why isn’t addiction considered medical?
The underlying problem is that for nearly the entire history of medicine, addiction was never considered a medical disease, even though the consensus in the neuroscience of addiction field the past few decades is that addiction absolutely is a medical disease, a disease of the brain. Public opinion and professional medical attitudes alike need to change because without proper diagnosis of a disease there cannot be proper treatment.
The historical stigma of addiction
Historically, a drug addict was considered someone that was weak, lacked a strong will, or was morally inferior. These assessments are not true but the stigma around addiction still persists today.
The textbook definition of addiction (according to Wikipedia) is “is a state characterized by compulsive engagement in rewarding stimuli, despite adverse consequences.” But drugs of abuse (nicotine, cocaine, marijuana, alcohol, heroin, oxycodone, etc.) are not magic: they are physical substances that have a physical effect on many systems especially control centers in the brain.
The function of an addict’s brain has quite literally been changed as a result of the drug use. And with repeated drug use, the function of the brain changes to such a degree that an addict’s behaviors and motivations—what you might call “will power”—have changed. An addict is not “morally weak” but suffering from an illness of the brain and in need of compassion and proper medical treatment.
So why is addiction a disease of the brain and what does this even mean? What is the scientific evidence that supports this idea? How do drugs hijack the brain and change behavior? Obviously books have been written on this subject but I’ll provide a discrete yet thorough overview. But to begin this discussion let’s start from the bottom up.
What is the brain?
Before we can understand how drugs affect the brain or hijack the brain’s functions, it’s important to understand what the brain even is.
So we all know that our body is made up of organs: heart, lungs, intestines, etc. and those organs are responsible for carrying out different jobs that keep us alive. Just as the lungs are responsible for absorbing oxygen and eliminating carbon dioxide, and the heart is responsible for moving blood about the body, the brain’s functions involve regulating every other system in the body.
We were taught since we were young that the brain is the control center of the body, which is one of those phrases that is technically true but doesn’t really offer much insight. By control center, we mean that the brain controls how our organs function (breathing, heart rate, muscle movement, etc.). However, the brain does more than just regulate the essentials. As neuroscienctific research has shown, every thought, feeling, emotion, memory, opinion, idea, action, or choice you’ve ever made or ever will make is the result of processes that occur in the brain. This may sound controversial but the function of the brain is the mind.
The brain = Our behavior
Phrased another way, the sum total of all the brain’s internal processes (emotions, thoughts, etc.) is our behavior (choices, actions, etc). Granted that how the brain achieves these incredible feats is what the neuroscience field is trying to learn but a wealth of evidence already suggests this to be true.
For example, human patients that have a lesion or damage in a particular region of the brain lose the function or behavior that brain region is responsible for (see this review article [1] as an example of this type of research). Or another example is Alzheimer’s disease, which is a brain disease that involves progressive loss of long-term memories and other cognitive process. Or Parkinson’s disease involves a loss of control over voluntary movements. In both of these cases, clearly brain function is important for both memory and voluntary movement because with loss of brain function, those processes are lost [2].
The point is that ultimately the brain is responsible for our behavior. I have only presented a few examples and much more evidence exists but let’s just assume that I’m right and agree that the function of the brain is indeed responsible for our behaviors (no neuroscientist would dispute this fact). But how does the brain control our behavior?
How does the brain work?
As I said, the brain is an organ and like every other organ in the body, the brain is made up of cells, and just as heart cells are responsible for heart function and lung cells for lung function, brains cells are responsible for brain function. We call the main type of brain cells neurons (there are many other types of brain cells called glial cells but I won’t be discussing those in this post). The human brain contains some 86 billion neurons that make trillions of connections with each other [3].
Neural communication
The primary function of a neuron is to conduct an electrochemical signal to other neurons. When a neuron “fires”, it means that it conducts an electrical signal called an action potential along its length, called the axon.
The end of an axon of one neuron connects with another neuron at special branches called dendrites and the junction between the two is called a synapse. When an action potential reaches the end of an axon it results in the release of special “communication” molecules called neurotransmitters into the synapse.
When neurons “talk to each other” it means one neuron has fired an action potential and released neurotransmitters onto another neuron. Once the second neuron receives the neurotransmitter one of two things can happen:
- it can fire it’s own action potential or
- it can be prevented from firing
Drugs cause neurons to release neurotransmitters
It is very important to understand the concept of how neurons communicate with each other because drugs basically work by causing specific neurons to fire and release neurotransmitters.
Neurons are able to “talk with” hundreds or even thousands of different neurons. And there are many different types of neurons that have different properties and release different neurotransmitters. Indeed, some neurons are so different that we can classify them into distinct parts of the brain or brain regions.
For example, an important brain region at the core of addiction is the ventral tegmental area (VTA) and VTA neurons release the neurotransmitter dopamine onto neurons in another brain region called the nucleus accumbens (NAc) but I’ll talk more on the VTA, NAc, and dopamine later [4].
In the end, the astonishingly complex and vast network of neurons firing and releasing neurotransmitters onto each other comprises the brain and all of its functions.
How do drugs affect the brain?
Drugs are chemical substances, specific molecules that have specific properties and actions. Once a drug enters the body it interacts with and affects a particular type of molecule that exists in brain cells.
For example, cocaine blocks protein molecules call monoamine transporters. These monoamine transporters exist on the surface of neurons and are responsible for “clearing” neurotransmitters like dopamine from the synapse. Cocaine blocks this function so when a person takes cocaine, the transporter molecule can’t work and extra dopamine accumulates in the synapse, which causes neurons that respond to dopamine to activate (such as neurons in the NAc) [5].
Opioids are also able to change how neurons function. Opioids such as morphine, oxycodone, and heroin all act on and activate (instead of inhibit) a different protein molecule called the mu opioid receptor (MOPR). Specifically, opioids activate MOPR that are present on VTA neurons and when this happens a chain reaction occurs that results in some VTA dopamine neurons firing and releasing dopamine onto NAc neurons [6].
Drugs = Dopamine release from VTA to NAc neurons
Other drugs of abuse affect different molecules but they all have one thing in common: all addictive drugs (nicotine, alcohol, cocaine, heroin, oxycodone, marijuana, methamphetamine, etc.) increase dopamine release from VTA neurons to NAc neurons [7]. Dopamine release from VTA to NAc is the primary circuit in the brain the controls the perception of pleasure or reward. Not only do drugs of abuse increase dopamine, but also fatty food, sugar, gambling, and even sex do the same thing [8].
One theory of addiction is that dopamine neurons become desensitized as a result of chronic drug use, thus the pleasure a person experiences is reduced in the absence of the drug and more drug needs to be taken in order to restore the addict’s mood to “normal” [9]. This may also explain why increased amount of drug needs to be taken over time in order to feel the same “high”.
What is the evidence that drugs cause change brain function?
Now that we know how drugs physically alter the function of neurons, especially neurons that release and respond to dopamine, what does this mean for the brain as a whole? How does altered neuronal function result in the compulsive drug taking behavior that is the hallmark of addiction?
Neuroimaging studies of the brain on drugs
Some of the best evidence we have comes from neuroimaging studies. Neuroimaging techniques such as positron emission tomography (PET) functional magnetic resonance imaging (fMRI) are able to monitor how much activity is going on in a particular brain region in a living person.
For example, compared to control patients, brains of cocaine addicts have reduced sensitivity of dopamine neurons in the striatum (a region of the brain that includes the NAc)[10]. This suggests that the function of these neurons has been altered and this may help drive increased usage of cocaine by using the drug to return dopamine activity, and mood, to normal.
Another important region that acts on the NAc is the prefrontal cortex (PFC). If the VTA is the accelerator that drives drug use, then the PFC is the brakes that curb it. Neuroimaging data has shown PFC function to be reduced in addicts [11]. This finding suggests that the “brakes have been cut” and the drive to take drugs is dominating the addicts behavior; they have physically lost their self-control or “will power”! Neuroimaging data has also revealed changes in many other brain regions such as the hippocampus and amygdala.
A great deal of evidence has been gathered from animal behavioral studies. One powerful technique that the lab I work in at the Rockefeller University (and many other labs) use is called intravenous self-administration or just self-ad for short.
Self-ad experiments and molecular changes
In a self-ad experiment, a mouse or rat is surgically implanted with a catheter that is inserted into its jugular vein. The animal is than hooked up to an experimental device called the operant test chamber. In the test chamber the animal has a choice between two levers (or nose-holes in some chambers): the active lever will deliver a dose of the drug directly into the animal’s bloodstream while the inactive lever will do nothing.
Animals that are allowed to self-administer drugs like cocaine or heroin will repeatedly push (or nose-poke) the active, drug lever and if you run the experiment once a day over a few weeks, the animals will self-administer increasing amounts of drugs. We call this, “escalation” and there is a similar type of drug-taking behavior found in humans. Importantly, control animals that are delivered saline (a salt solution) do not repeatedly lever press or escalate, which shows that the rodents “lever-pressing behavior” or desire to receive the drug, is because of the drug itself.
We can use self-ad studies and other behavioral experiments to analyze the effects of drugs on the brain of rodents and other animals to a level of precision that is simply not possible in humans. After a behavior experiment, the rodent is sacrificed, its brain is removed, and specific brain regions, such as the NAc and VTA are dissected out. We can then use the removed brain tissue to run molecular analyses.
One of our most important molecular changes that we can measure is gene expression. In brief, when any neuron (or any cell) responds to a stimulus, new protein molecules may be needed to respond to this change, which means DNA needs to transcribe more RNA so that it can be translated into more protein. An increase in RNA is called upregulated gene expression.
A critical gene that is upregulated after a drug administration behavior experiment for a variety of different drugs is dynorphin. Dynorphin acts at the kappa opioid receptor (KOPR), a gene that is related to MOPR but acts in the exact opposite way, that is to say activation of KOPR suppresses dopamine release from VTA neurons whereas activation of MOPR increases it. Repeated activation of the dynorphin/KOPR system can cause long-lasting alterations in the dopamine system [12].
Indeed, one hypothesis about how addiction develops involves the increase of KOPR activity in the VTA/NAc circuit of addicts, which causes the feelings of dysphoria (unhappiness or dissatisfaction) and anhedonia (lack of pleasure) that occur during abstinence and drives the desire to take the drug again in order to relieve these negative feelings [13].
These are just a few examples of the concrete molecular changes that occur in the brain as a result of repeated drug use. Future blog posts may discuss more of this evidence in detail.
What is a disease?
Ok, just to summarize the discussion up to this point: we now know that the brain is made of neurons and drugs change the molecular function of these neurons, which results in a long-lasting change in behavior. But does this mean that addiction is disease?
You may or may not have thought about this in this way, but the entire modern medical profession is based on a standard way of treating illness: the medical model of disease. The model is simple to understand: illness happens because something (bacteria, virus, a genetic mutation, a poison, etc.) affects a particular organ (actually usually specific molecules within the cells of those organs), causing it to not work properly and resulting in the symptoms of the disease. Therefore, if you fix either the cause of the problems to the organ (or in some cases the problems themselves), you eliminate the symptoms and cure the disease.
Does the disease model work for addiction?
If something changes how the brain operates (like drugs do), then our thoughts and behaviors would also be changed. If this change results in negative behaviors or thoughts, you could think of the brain as suffering from a disease. Now let’s apply this thinking to the medical model of disease.
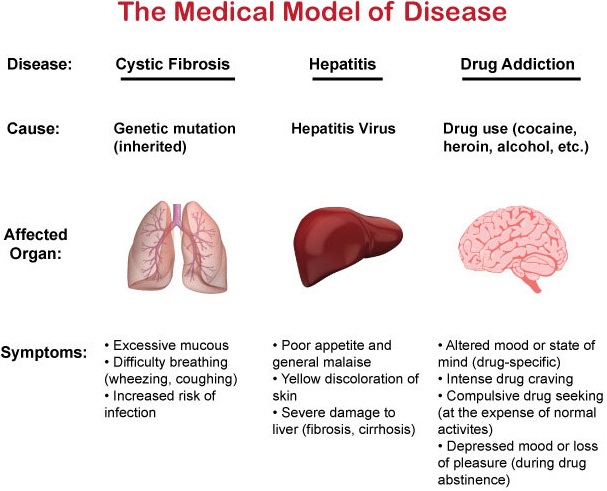
The figure below compares three different diseases in the context of the disease model: cystic fibrosis, hepatitis, and drug addiction.
For cystic fibrosis, the cause of the disease is a genetic mutation that you inherit from your parents. The organ the mutation affects is the lung. The mutation causes the lung to produce more mucous which makes breathing more difficult (the symptoms).
For hepatitis, a virus, the hepatitis virus, causes the disease. The virus specifically infects the liver, the affected organ, which damages it and causes a loss of appetite and malaise, and can lead to yellow discoloration of the skin or more severe liver damage (the symptoms).
For drug addiction, the cause of the disease is drug use. The drugs act on brain cells (neurons), which changes how they work which makes the brain the affected organ. The change in brain function results in the drug-specific effects that you experience right away, while repeated use results in cravings, drug-seeking behavior, and even withdrawals (all of these are symptoms because a change in behavior is considered a symptom).
Based on this this in combination with all of our knowledge about how drugs change brain function and behavior, we can conclude that addiction is indeed a disease.
Conclusion
I hopefully have provided you with enough evidence to convince you that the brain controls behavior and this function is because of neurons that communicate with each other through neurotransmitters. We know that drugs directly affect neurons and cause long-term changes in the functions of neurons, which changes behavior. And we know that this process is parallel to every other disease. At this point, but I would like to return to the behavior aspect briefly just to conclude our discussion.
Addiction is like extreme hunger
The brain/mind/behavior interaction can be complex and difficult to understand because it certainly seems like we are in control of all our actions and how can some random chemical process change how one thinks?
Conceptualize it a different way using this example: if you are energy deprived, the brain will perceive the sensation of “hunger” which will result in mental processes such as motivation and decision-making that will result in a person performing a behavior. In this case, the behavior is eating food, which ameliorates the problem (lack of energy) and relieves the negative sensation (hunger).
In the addicted brain, one of the problems is lack of dopamine, the sensation the brain perceives is dysphoria, anhedonia, and a craving for the drug and the behavior is taking the drug. And with time, these changes can become so severe that drug taking becomes the dominant behavior.
You’ve heard of people so desperately hungry that they’ve resorted to cannibalism? Drugs are so powerful and the craving so intense, drug-taking behavior can become the only goal, even if achieving that goal hurts the addict or others around them. And these changes are the disease state and why addiction is a disease.
One thing I did not discuss is the medical treatments available to help stop this process, but that discussion will be reserved for a future post. Stay tuned for more.
Reference Sources:
-
Szczepanski SM, Knight RT. Insights into human behavior from lesions to the prefrontal cortex. Neuron. 2014;83(5):1002-18.
-
Hirao K, et al. Molecular imaging of neuropsychiatric symptoms in Alzheimer’s and Parkinson’s disease. Neuroscience and biobehavioral reviews. 2015;49:157-70.
-
Azevedo FA, et al. Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled-up primate brain. The Journal of comparative neurology. 2009;513(5):532-41.
-
Russo SJ, Nestler EJ. The brain reward circuitry in mood disorders. Nature reviews Neuroscience. 2013;14(9):609-25.
-
Ritz MC, et al. Cocaine receptors on dopamine transporters are related to self-administration of cocaine. Science. 1987;237(4819):1219-23.
-
Fields HL, Margolis EB. Understanding opioid reward. Trends in neurosciences. 2015;38(4):217-25.
-
Nutt DJ, et al. The dopamine theory of addiction: 40 years of highs and lows. Nature reviews Neuroscience. 2015;16(5):305-12.
-
Volkow ND, et al. Food and drug reward: overlapping circuits in human obesity and addiction. Current topics in behavioral neurosciences. 2012;11:1-24.
-
Volkow ND, Morales M. The Brain on Drugs: From Reward to Addiction. Cell. 2015;162(4):712-25.
-
Volkow ND, et al. Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature. 1997;386(6627):830-3.
-
Goldstein RZ, Volkow ND. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nature reviews Neuroscience. 2011;12(11):652-69.
-
Butelman ER, et al. kappa-opioid receptor/dynorphin system: genetic and pharmacotherapeutic implications for addiction. Trends in neurosciences. 2012;35(10):587-96.
-
Koob GF, Le Moal M. Addiction and the brain antireward system. Annual review of psychology. 2008;59:29-53.








Related Posts